| property | value |
|---|---|
| Common names | DiPT |
| Substitutive name | N,N-Diisopropyltryptamine |
| Systematic name | 3-[2-(Diisopropylamino)ethyl]indole |
| Psychoactive class | Psychedelic |
| Chemical class | Tryptamine |
n,n-diisopropyltryptamine (also known as dipt) is a lesser-known psychedelic substance of the tryptamine class. it is related in structure to psychedelic tryptamine compounds like 4-ho-dipt, 5-meo-dipt, and mipt.
the first human trials of dipt were undertaken in 1975 by alexander shulgin, who would co-author and publish a paper detailing its synthesis and human psychopharmacology in 1981. a summary of the synthesis and reports of human use is included in shulgin’s 1997 book tihkal (“tryptamines i have known and loved”). it has been sold online as a research chemical.
while most psychedelics are known for producing vivid visual distortions, dipt’s effects are reported to be primarily auditory in nature. anecdotal reports have described it causing a downward shift in perceived pitch and other unusual distortions. it has been suggested that dipt may have value in neurological research due to its unique effects on auditory perception. additional subjective effects of dipt include a body high, moderate euphoria, dizziness, and nausea.
very little data exists about the pharmacological properties, metabolism, and toxicity of dipt, and it has a limited history of human use. it is highly advised to use harm reduction practices if using this substance.
chemistry
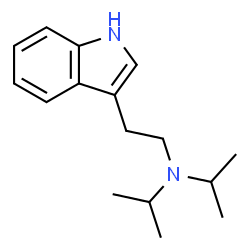
dipt, or n,n-diisopropyltryptamine, is a synthetic indole alkaloid molecule of the tryptamine class. tryptamines share a core structure comprised of a bicylic indole heterocycle attached at r3 to an amino group via an ethyl side chain.
dipt also contains two isopropyl groups bound to the terminal amine rn of its tryptamine backbone. isopropyl groups are a three carbon (propyl) chain bound at the middle carbon to the chemical structure. dipt has substituted analogues such 5-meo-dipt.
dipt is analogous to dmt, containing two isopropyl groups bound at rn instead of the two methyl groups found in dmt.
pharmacology
the psychedelic effects of dipt are believed to come from its efficacy at the 5-ht2a receptor. however, the role of these interactions and how they result in the psychedelic experience remains subject to on-going scientific investigation.


Reviews
There are no reviews yet.